Open Source Drug Discovery

Drug discovery is challenging, lengthy, and extraordinarily expensive. All companies focus on making money and drug companies are no different. They spend their research and development budgets focusing on diseases that affect the affluent world population. As a consequence diseases such as malaria and tuberculosis that are abundant in sub-Saharan Africa and India are left unstudied by industry. Despite the omission by drug companies, other efforts are underway to aid in drug discovery for these diseases.
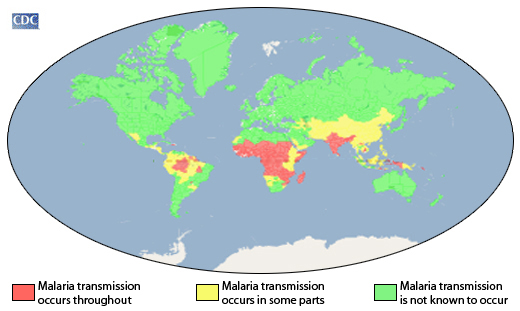
Just to hash out a few details of where the problem arises– the Center for Disease Control (CDC) states that 35 countries (30 African and 5 Asian) account for 98% of the malarial related deaths. Similarly, in 2010, 8.8 million people became sick with tuberculosis (TB) of which 82% of the cases lived in 22 TB ridden countries. These diseases do exist in other parts of the world, but result in significantly less deaths.
Despite the lack of interest from drug companies for treating these infectious diseases, other organizations have picked up the slack. The Bill & Melinda Gates Foundation has contributed significant funds to these poverty ridden populations in the form of care, research, and vaccines. Much of their focus is on HIV/AIDS treatment in addition to other conditions including malaria and TB. However, while reading Science I recently came across an open source drug design initiative based out of India.
A few years ago, Samir Brahmachari launched the Open Source Drug Discovery (OSDD) network. The initiative began in 2008 and set out to combat India’s leading cause of death, TB. The initial $12 million of seed money was provided by the Indian government and that has led to 5500 participants in 130 countries. So has this global network of researchers provided any results? Their first goal was to sequence the TB genome and the task was accomplished in a mere four months by 500 volunteers. Since then they utilized this information and have determined two viable drug candidates that are currently being tested. Following the principles of OSDD, the data from their clinical trials are open for all to see. It is too soon to tell if the drugs will be successful, but if so they will be on the market as generic drugs.
This approach could produce affordable health care and treatments for many without.




I’d love to see this work and inspire future efforts. It could even provide a new model in the U.S.–publicly funded universities could produce research, with the government holding the patent initially until sales had recouped the initial investments, and then moved to open-source promptly. The government could easily recoup the expenditure, and even generate a small profit, on a fraction of the price that Big Pharma typically charges.
Another way I always thought might work it’s to tell drug companies that if you want to sell your products in this country you have to do research into “orphaned” diseases.
And make it proportional, for every, say, 10 drugs you have on the market you get another disease to research (not their choice). If you develop a helpful drug you get a pass on 1 orphaned drug for 5 years as incentive to actually try.
Oh and the drug itself is not patentable.
Just call it the price of doing business.
Probably makes me a socialist commie fascist our something.
Bad capitalist, bad!